What is the Boyle's Law ?
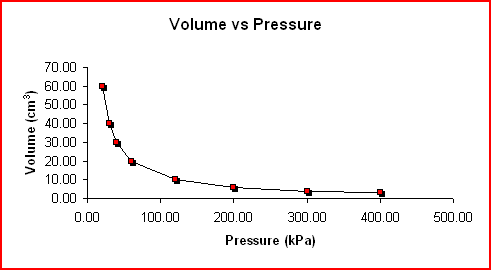
- Boyle's law states that the volume of a fixed amount of gas held at a constant temperature varies inversely with the pressure.
- Volume (v) and pressure (p) are the two variables that relate
- The relationship between the two variables is inverse.
- As the pressure increases, the volume decreases.
- The constant is the temperature.
(The Image located at the top of the page is the formula used for the Boyle's law)
- Volume (v) and pressure (p) are the two variables that relate
- The relationship between the two variables is inverse.
- As the pressure increases, the volume decreases.
- The constant is the temperature.
(The Image located at the top of the page is the formula used for the Boyle's law)
(This graph shows that as you go across the x-axis the pressure is increasing. As you look at the line you see the line going down, representing, as pressure increases the volume decreases.)